Project Lead(s): Matt Ratto
Issue
Traffic accidents, civil unrest, landmines, natural disasters, congenital conditions, and diseases such as polio and cerebral palsy have left hundreds of thousands of endemically poor children in need of quality prostheses.
According to the World Health Organization (WHO), approximately 30 million people in low-income countries require prosthetic limbs, braces or other assistive devices. However, only 5–15% of the people who need these devices have access to them.
Obstacles to access include a shortfall of 40,000 orthopaedic personnel, time-consuming and labour-intensive production methods that require an average of five days to produce a prosthesis, and significant costs to patients, especially those who travel vast distances for treatment.
Without prostheses, children with disabilities face social ostracism and a dramatically reduced quality of life, which often limits their ability to access education and future employment.
Solution
With too few orthopaedic technologists to meet the demand for assistive devices, innovative uses of 3D printing technology offer a way to drive down prosthetic production times, while increasing the comfort and fit of the device for users.
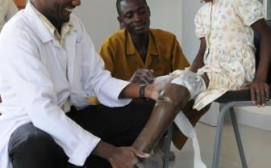
3D PrintAbility is a Canadian innovation that combines 3D scanning, modelling and printing technologies to produce custom prosthetic sockets – complex components that connect an artificial limb to the patient’s residual limb. Initiated by cbm Canada in 2013, the 3D PrintAbility concept was developed by researchers at the University of Toronto and Autodesk Research, and supported by Phase I funding from Grand Challenges Canada.
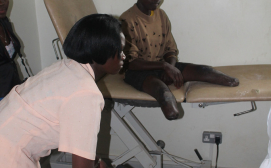
3D PrintAbility is a digital design and production toolchain, complete with custom software and affordable hardware (scanner, laptop and 3D printers), that produces below-the-knee prosthetic sockets more quickly than is possible with conventional plaster cast production methods.
3D PrintAbility builds on the skills and expertise of orthopaedic personnel, allowing them to minimize time on manual production and maximize time on decisions about device design, fit and patient care.
Outcome
3D PrintAbility’s first clinical trial tested its ‘proof-of-concept’ at Comprehensive Rehabilitation Services in Uganda (CoRSU) from January to July 2015, with 32 child/youth participants (4–25 years of age).
The trial validated 3D PrintAbility in several key ways: orthopaedic personnel were trained to produce biometrically accurate 3D-printed sockets; production time was cut by 70–80%; patients were fitted with custom-designed prostheses in less time (1.5 days rather than 5 days); and the toolchain was rigorously tested and its fit in a developing-world setting demonstrated. Feedback from the trial informed toolchain improvements, further materials testing and product expansion into ankle foot orthoses (braces).
In 2015, cbm Canada created Nia Technologies Inc. (Nia), a non-profit social enterprise, to further develop, scale and deploy 3D PrintAbility in low- and middle-income countries.
Nia received Phase II funding from Grand Challenges Canada to scale the technology, with matching funding from Autodesk Foundation, Google.org, Stronger Philanthropy and others.
Funding will enable Nia to build capacity, conduct ongoing R&D and additional clinical trials, transfer knowledge to more orthopaedic personnel, create an open-source, online peer review system for 3D PrintAbility designs, and to study deployment models.