Arbutus 0742-05
Project Lead(s): Florin Gheorghe
A novel medical device revolutionizing orthopaedic surgery in the developing world will be launched in new markets through Grand Challenges Canada’s support, allowing hospitals to improve infection control and help thousands of patients to avoid permanent disability. This product is now FDA listed in the US and has treated over 10,000 patients across 14 countries.
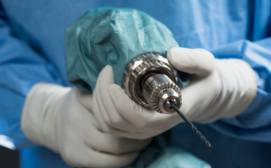
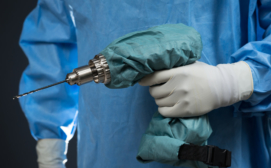
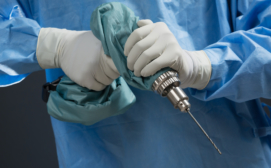
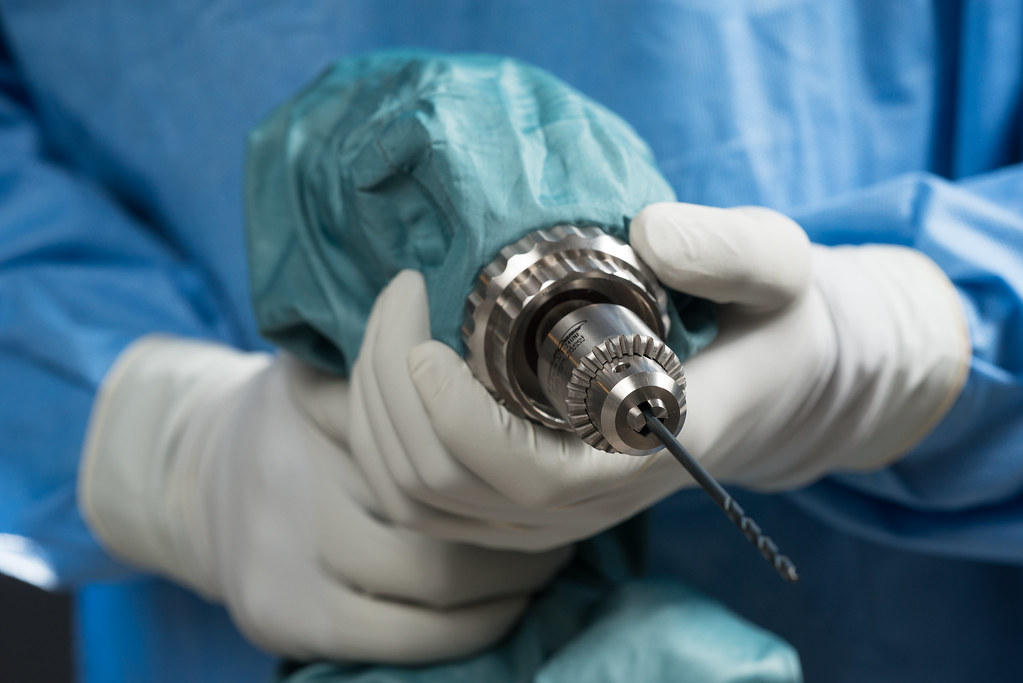
The Drill Cover, developed by Arbutus Medical, a Vancouver-based global health tech start-up, is surgical grade, sterilizable, reusable, waterproof, and blood and pathogen resistant.
It envelops a low-cost hardware store drill, enabling its safe use for bone surgery – presenting a significantly more accessible alternative to surgical drills priced up to $30,000.
In low- and middle-income countries, basic surgical care has the potential to play a major role in reducing the burden of injuries. However, 5 billion patients do not have access to safe surgical care. Lack of access to affordable care is a significant barrier for many hospitals trying to provide surgical care to thousands of patients.
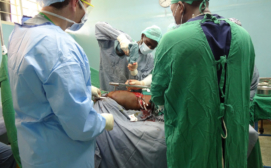
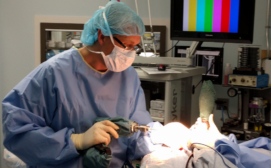
Surgical drills are often not available in resource-constrained areas, forcing hospitals to resort to inefficient and dangerous “hand-crank” drills, or improvise with nonsterile construction drills.
In clinical tests comparing the Drill Cover innovation to hand drills, also funded by Grand Challenges Canada’s Stars in Global Health program, the Drill Cover shortened surgical time by nearly 30 minutes per case, while surgeons reported fewer problems during bone drilling tasks.
With new transition-to-scale funding from Grand Challenges Canada and partners, Arbutus has completed remaining product refinements, successfully achieved FDA listing in the US, and is currently scaling the product across East Africa and India, while developing new products for tools such as surgical saws.
Arbutus’ primary focus is on hospitals and surgeons in low- and middle-income countries, but has complementary opportunities with disaster/conflict relief organizations, veterinary surgeons, and military hospitals.
Arbutus is collaborating closely on scaling this product with distribution partners such as Crown Healthcare Africa, working on market strategy implementation in India with Intellecap, and is supported through mentorship by Edmonton-based medical device company priMED Medical Products