Project Lead(s): Carlos Delgado Bocanegra
Issue
Worldwide, almost 10% of newborn babies (13.6 million annually) require some help to begin to breathe during the first 30 seconds of life.
If trained health personnel are available, they provide tactile stimulation through warming, positioning and drying. When additional support for neonatal resuscitation is needed, the World Health Organization recommends use of a self-inflatable bag attached to a facial mask.
However, the bag and mask have many constraints: trained personnel are required, there is ample variability in performance and devices are scarce, especially when birth occurs outside the hospital in middle- or low-income countries.
Solution
The objective of the project was to develop a portable, user-friendly device that is cheaper than conventional bag-and-mask devices, and that can generate sufficient airflow and pressure to stimulate the onset of spontaneous breathing in newborns.
The principal hypothesis behind the project was that a respiratory stimulus is as effective as a tactile stimulus in triggering spontaneous breathing, even before additional requirements, such as use of the bag and mask.
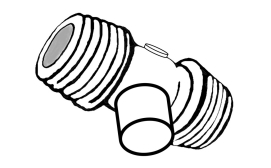
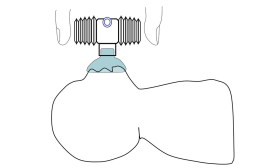
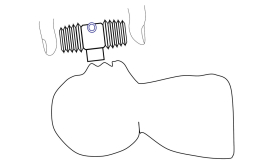
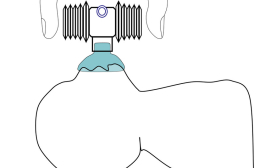
The prototype developed is a device with a closed centre and bellows in two sizes (8 and 12 ml). The bellows are attached in a lateral position, using a nut and screw mechanism. This prototype generates airflow and pressure, pushing air, using only two fingers over the bellows.
The team conducted laboratory tests using the prototype attached to a Flow-Gas-Analyzer (VT-305, Fluke Medical, U.S.A.)
Outcome
The prototype device was able to achieve values of 5 to 7 lt/min for Peak Inspiratory Flow and 6 to 8 cmH2O for Peak Inspiratory Pressure. Those values are small enough to minimize the possibilities of lung injury.
The team trained 58 healthcare personnel (51 nurses and 7 physicians) in neonatal resuscitation and all participants were invited to test the device using the Flow-Gas-Analyzer.
These results, including laboratory tests, will be presented as a research report at a later stage. No clinical study was conducted, due to new restrictions on testing involving children in Peru.
Before the prototype can be scaled for commercial purposes, it will require an assessment of efficacy and safety of respiratory stimulus in a pivot study and/or clinical trial. The team has contacted Partners in Health (PIH) to determine their interest as potential partners for prototype validation and scaling.