Project Lead(s): Holger Mayta
Issue
Classical swine fever (CSF) or hog cholera is a highly contagious and often fatal disease in pigs.
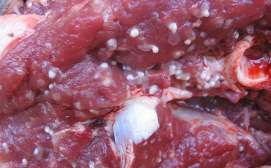
Porcine cysticercosis is a parasitic tissue infection caused by larval cysts of Taenia solium (pork tapeworm) that makes pork measly, causing major economic loss among pig farmers. The cyst also causes human neurocysticercosis, the major cause of adult onset seizures in most low-income countries.
Solution
The present project aimed to develop a cheaper bivalent vaccine that will protect pigs against both hog cholera and T. solium cysticercosis.
The Tsol18 genes (Tsol18 is a protein derived from T. solium that is expressed as a recombinant peptide in E. coli fused to glutathione S-transferase (GST) and has shown close to 100% efficiency in protecting pigs after two doses) and E2 genes (E2 is a glycoprotein that has shown high efficiency in protecting pigs against hog cholera and is commercially available) were cloned directionally into a pFastBac1 vector.
The gene sequence was optimized for baculovirus expression.
Both proteins were histidine tagged and, to ensure availability of individual peptides, a self-cleaving short peptide (T2A) was inserted between the two genes.
Protein expression optimization at low scale was initially performed at the University of Maryland Baltimore County (UMBC); after optimization, a scale-up production was commercially performed.
A vaccine trial was performed in different field sites in Peru.
Outcome
The results showed that a bivalent vaccine for both classic swine fever and porcine cysticercosis shows great promise in controlling the diseases.
Both Tsol18 and E2 were successfully expressed in the Baculovirus expression system, and were present in the cellular pellet as individual proteins, as corroborated by Western blot using anti-his tag antibodies and sera from naturally infected pigs.
After the second vaccine dose, the antibody levels against Tsol18 were significantly higher in the vaccinated pigs than in the control pigs. However, the antibody levels were lower than those observed when GST-Tsol18 expressed in E. coli was used in a vaccine.
The antibody levels against E2 were higher in the vaccinated group than in the non-vaccinated group, but lower than those reached when life attenuated vaccine was used.
When vaccinated pigs were challenged, the vaccine tested in this study failed to protect pigs from acquiring cysticercosis. However, the number of cysts present in vaccinated pigs were lower than in non-vaccinated pigs.
Overall, the vaccine tests in this study confer an antibody response lower than those observed with the individual vaccines that were not efficient in protecting pigs against T. solium cysticercosis.
Collaborative work is ongoing with the main vaccine producer in Peru.
The team has synthetized a second vaccine with similar characteristics, based in a second T. solium protein candidate, that will be tested once additional funds are available.